Rapid In Vivo Antibody Discovery
Accelerating Biotherapeutics with Innovation
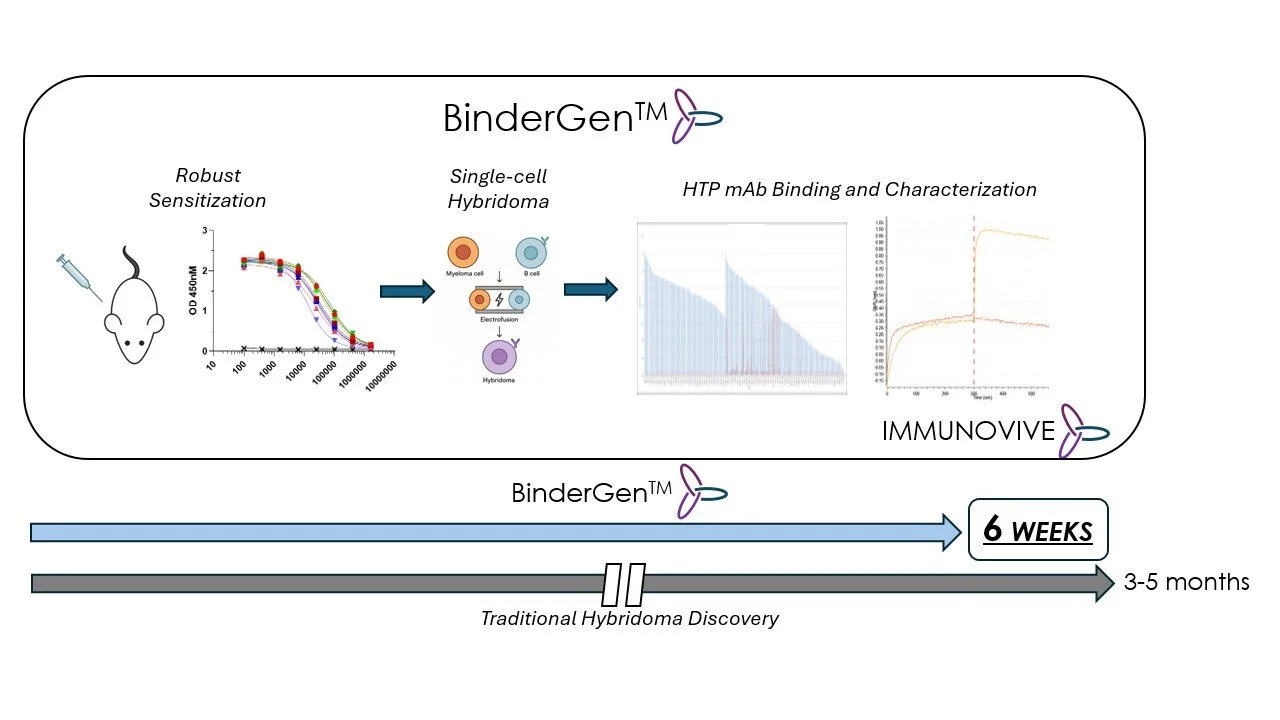
The BinderGenᵀᴹ Platform
De Novo Monoclonal Antibody Discovery at Unprecedented Speed
In 6 weeks, Immunovive’s In vivo BinderGenᵀᴹ platform delivers:
· Diverse panel of target-specific affinity-ranked monoclonal antibodies
· Natively paired VH/VL sequences
· Subnanomolar affinity or better
· Broad repertoire diversity
· >70% reduction in discovery timelines
· Validated in virtually all rodent systems including humanized, knockouts, conditional, inducible, and transgenics.

Our Mission
Our mission is to accelerate the discovery and development of immunotherapeutics. Our BinderGenᵀᴹ lead discovery platform generates diverse in vivo-matured monoclonal antibody target binders in 6 weeks built to empower downstream preclinical drug development processes including target validation, proof-of-concept, lead identification and optimization, critical reagents, diagnostics, and provide high-quality substrate for multimodality engineering and AI/ML datasets.
Our Team
Devin E. Turner, Ph.D.
Founder and CEO
Rashed Abdullah, Ph.D., PMP
Associate Director of Operations and Business Development
Tom Daly, Jr., M.Sc.
Scientist